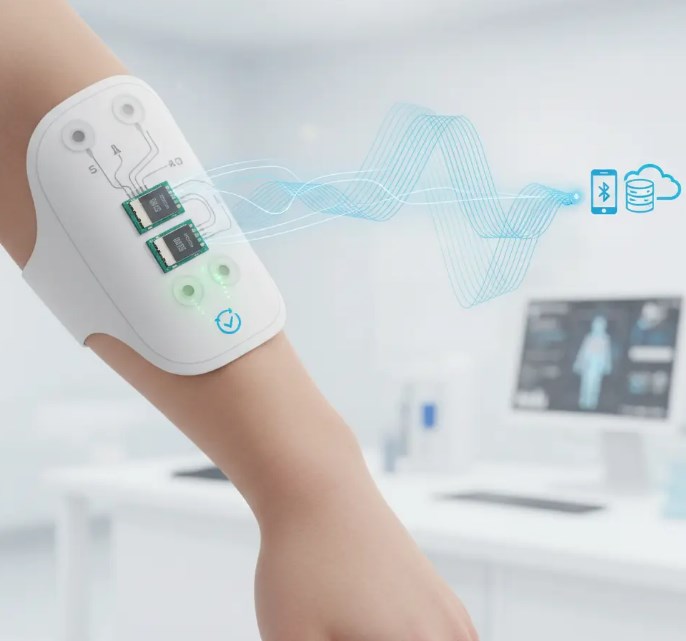
Are you struggling with strict compliance, reliability and miniaturization in wearable medical device PCBA? EBest solves your core pain points with ISO 13485 certification, high-precision assembly, and full‑cycle prototype to mass production support. We provide reliable, regulated, and fast wearable medical device PCBA solutions for medical, aerospace and industrial applications. Choose EBest as your trusted partner to accelerate approval and launch safely.

Why Choose EBest for Wearable Medical Device PCBA Solutions?
EBest is your top partner for wearable medical device PCBA with full ISO 13485 compliance and 19+ years of medical electronics experience. We deliver reliable, regulation-ready assemblies that speed up your certification and market launch, while ensuring safety and stability for patient‑used devices.
We specialize in high‑precision miniaturized assembly ideal for wearable designs, including flex PCB, ceramic PCB, 01005 components, and 0.25mm pitch BGA. Every unit goes through 100% full inspection, strict traceability, and controlled production to meet the strictest medical quality standards.
We offer one-stop wearable medical device PCBA solutions covering prototype to mass production, with fast 1–5 day lead times and 24-hour expedite service. Our full turnkey support simplifies your supply chain and reduces project risks, making us the most reliable choice for your medical wearable manufacturing.
Common Pain Points in Wearable Medical Device PCBA & Our Solutions
- Strict medical regulatory compliance (ISO 13485) required
- Pain Point: Many manufacturers lack medical-grade certifications and cannot support regulatory submissions for wearable medical devices.
- Solution: EBest is ISO 13485:2016 certified with full traceability, controlled production, and complete documentation for FDA/CE compliance.
- Miniaturization & high-density assembly challenges
- Pain Point: Wearable devices need small size, but fine-pitch BGA and tiny components are hard to assemble stably.
- Solution: We support 01005 components and 0.25mm BGA pitch with high-precision SMT lines and 3D/AOI/X-Ray inspection.
- Flexible PCB reliability & bending durability
- Pain Point: Flex PCB easily breaks or fails under repeated body movement.
- Solution: Professional flex & rigid-flex PCB assembly with optimized materials and processes for long-term bending stability.
- Unstable component supply & poor traceability
- Pain Point: Long lead times, fake components, or missing traceability records block medical certification.
- Solution: Stable medical-grade component sourcing with full lot traceability from raw material to finished goods.
- Slow prototype & long production lead time
- Pain Point: Slow development delays product launch and market entry.
- Solution: Quick-turn prototype support and 1–5 day standard lead time, with 24-hour expedite service for urgent projects.
- Lack of one-stop manufacturing support
- Pain Point: Cooperating with multiple suppliers increases risk, cost, and management workload.
- Solution: Full turnkey wearable medical device PCBA solutions including PCB fabrication, assembly, box build, and value-added services.
- Insufficient quality inspection for medical applications
- Pain Point: Inadequate testing leads to product failure and safety risks in patient-worn devices.
- Solution: 100% full inspection before delivery, including AOI, X-Ray, ICT, and functional testing.
Why EBest Is Your Trusted Wearable Medical Device PCBA Manufacturer?
Reasons why EBest is your trusted wearable medical device PCBA manufacturer:
- ISO 13485 Certified for Medical Devices: We hold official ISO 13485:2016 certification, fully compliant with global medical industry regulations to support your FDA, CE, and regulatory submissions.
- Proven Experience in Wearable Medical Electronics: 19+ years of specialized experience in wearable medical device PCBA, serving medical, aerospace, and industrial high‑reliability markets.
- Full Regulatory & Traceability System: Complete material and production traceability, strict quality documentation, and controlled manufacturing environments for medical safety.
- High‑Precision Miniature Assembly Capability: Support for 01005 components, 0.25 mm BGA pitch, flex PCB, rigid‑flex PCB, and ceramic PCB for compact, body‑friendly wearable designs.
- One‑Stop Turnkey PCBA Solutions: Full service from PCB fabrication, component sourcing, SMT/THT assembly, box build, to value‑added processes, simplifying your supply chain.
- Fast Prototyping & Volume Production Support: Quick‑turn prototypes, stable mid‑volume, and scalable high‑volume production with 1–5 day lead times and 24‑hour expedite service.
- Advanced Quality Inspection & Testing: 100% full inspection including AOI, X‑Ray, ICT, and functional testing to ensure zero defects for life‑critical wearable medical devices.
- Stable & Compliant Component Supply Chain: Reliable medical‑grade component sourcing with full traceability, avoiding counterfeit parts and ensuring long‑term project stability.
- Aerospace & Automotive Grade Standards: Certified with AS9100D, IATF 16949, ISO 9001, delivering the same high reliability used in aerospace and automotive applications.
- Environmental & Safety Compliance: Fully RoHS, REACH, and UL compliant, meeting global environmental and safety requirements for medical electronics.
- Professional DFM & Engineering Support: Free design for manufacturing reviews to optimize your design, lower risks, improve yield, and speed up mass production.
- Full Box Build & Final Assembly Services: Injection molding, CNC machining, sheet metal, pad printing, laser engraving, and finished product assembly for turnkey delivery.
Our ISO 13485 Certified Wearable Medical Device PCBA Services
EBest provides a complete suite for wearable medical device PCBA:
- PCB Fabrication: FR‑4, multilayer, metal‑core, ceramic, flex/rigid‑flex, high‑frequency.
- Component Sourcing: Full traceability, SMD, BGA, QFN, QFP, medical‑grade components.
- Assembly: SMT, THT, mixed, BGA, flex PCB, ceramic PCB, prototype, quick‑turn, full turnkey.
- Value‑added: Injection molding, CNC machining, sheet metal, box assembly, 100% inspection.

What Capabilities Do You Need for Wearable Medical Device PCBA?
| Capability Item | Specification |
| Placement Speed | 13,200,000 chips/day |
| Bare Board Size | 0.2×0.2 inch – 22×47.5 inch |
| Minimum SMD Component | 01005 |
| Minimum BGA Pitch | 0.25 mm |
| Max Component Assembly Area | 50×150 mm |
| Assembly Type | SMT, THT, Mixed Assembly |
| Component Package | Reels, Cut Tape, Tube, Tray, Loose Parts |
| Lead Time | 1–5 days; 24-hour expedited |
How to Ensure Reliability for Wearable Medical Device PCBA?
Reliability is non-negotiable for wearable medical devices, as they are used on the human body and provide critical health data. Below are the proven methods we use at EBest to deliver stable, long-lasting wearable medical device PCBA.
- Use medical-grade raw materials and biocompatible surface finishes: Only approved, traceable materials are used to avoid skin irritation, material degradation, or signal interference during long-term wear.
- Select flex / rigid-flex / ceramic PCB for body-friendly designs: These substrates resist bending, twisting, and perspiration, greatly improving durability for curved, portable wearable devices.
- Strict ESD-controlled and clean production environment: Static electricity and dust can cause hidden failures in medical electronics. Our fully controlled workshop prevents micro-damage to components and circuits.
- High-precision assembly for miniaturized components: Stable soldering for 01005 chips, 0.25mm BGA, QFN, and other fine-pitch packages ensures no cold joints, tombstoning, or short circuits.
- Full X-Ray, AOI, and SPI inspection: Internal solder joints (especially BGA) are checked layer by layer. Automated optical inspection ensures 100% visual and structural quality.
- ICT and functional testing for every board: Electrical performance and functional verification confirm each PCBA works as designed under real working conditions.
- Complete material and production traceability: Every batch of materials, process step, and test result is recorded to meet ISO 13485 requirements and support medical regulation audits.
- Thermal cycling and reliability verification: Simulate long-term use, temperature changes, and physical stress to avoid failure in real-world daily use.
- Controlled reflow soldering profile: Optimized temperature curves prevent PCB warping, component damage, and solder joint fatigue for long-term stability.
- Stable and certified component supply chain: We only use authentic, traceable electronic components to avoid counterfeit parts that cause sudden failures.

Prototype & Volume Support for Wearable Medical Device PCBA
We provide full-lifecycle manufacturing support for wearable medical device PCBA, covering early design validation, small‑batch trials, and large‑scale mass production. Our production system is built for medical regulation, flexibility, and speed, so you can launch products faster while maintaining consistent quality.
For prototyping and low‑volume production, we offer quick‑turn fabrication, DFM analysis, and rapid assembly to validate your design, materials, and functionality. This stage helps you identify risks early, optimize structure, and shorten the entire development cycle for your wearable medical device.
For mid and high‑volume production, we use automated assembly lines, strict process control, and full traceability to ensure stability and compliance. We maintain standard lead times of 1–5 business days and provide 24‑hour expedite service to support urgent project schedules and stable market supply.
BGA & High‑Density Assembly for Wearable Medical Device PCBA
High‑density assembly is the core technology of modern wearable medical device PCBA, as it enables smaller size, lighter weight, and stronger performance for body‑worn health products. EBest focuses on precision assembly for miniaturized, high‑value medical electronic circuits.
We support 0.25 mm fine‑pitch BGA, QFN, QFP, and 01005 ultra‑small components, which are widely used in compact wearable designs. Our high‑precision placement and optimized reflow soldering ensure stable, reliable solder joints without bridging, cold joints, or component damage.
We use 3D SPI, AOI, and X‑Ray inspection to verify every connection, including hidden BGA solder joints. Combined with flex/rigid‑flex PCB and ceramic PCB assembly, our high‑density solutions deliver excellent signal integrity, durability, and long‑term stability for critical wearable medical devices.
FAQ About Wearable Medical Device PCB Assembly
Q1: Do you hold ISO 13485 for wearable medical device PCBA?
A1: Yes, EBest is ISO 13485:2016 certified for medical electronics manufacturing.
Q2: Can you support prototypes and mass production?
A2: Yes, we cover quick‑turn prototypes to high‑volume production.
Q3: What flex and ceramic PCB capabilities do you have?
A3: We provide flex, rigid‑flex, and ceramic PCB design, fabrication, and assembly.
Q4: What’s the minimum BGA pitch you can assemble?
A4: We reliably assemble down to 0.25 mm BGA pitch.
Q5: How do you ensure component traceability?
A5: Full lot traceability from raw material to finished assembly.
Q6: What lead time can you offer?
A6: Standard 1–5 days; 24‑hour expedite available.
Q7: Do you provide box build and final assembly?
A7: Yes, full box build, injection molding, CNC, and sheet metal services.
Q8: Can you support medical regulatory submissions?
A8: Yes, we provide required process, test, and material documentation.
Get Your Wearable Medical Device PCBA Solutions from EBest
EBest delivers professional wearable medical device PCBA with ISO 13485 compliance, reliable quality, and fast delivery. For quotes, projects, or technical discussions, contact us at: sales@bestpcbs.com.


